 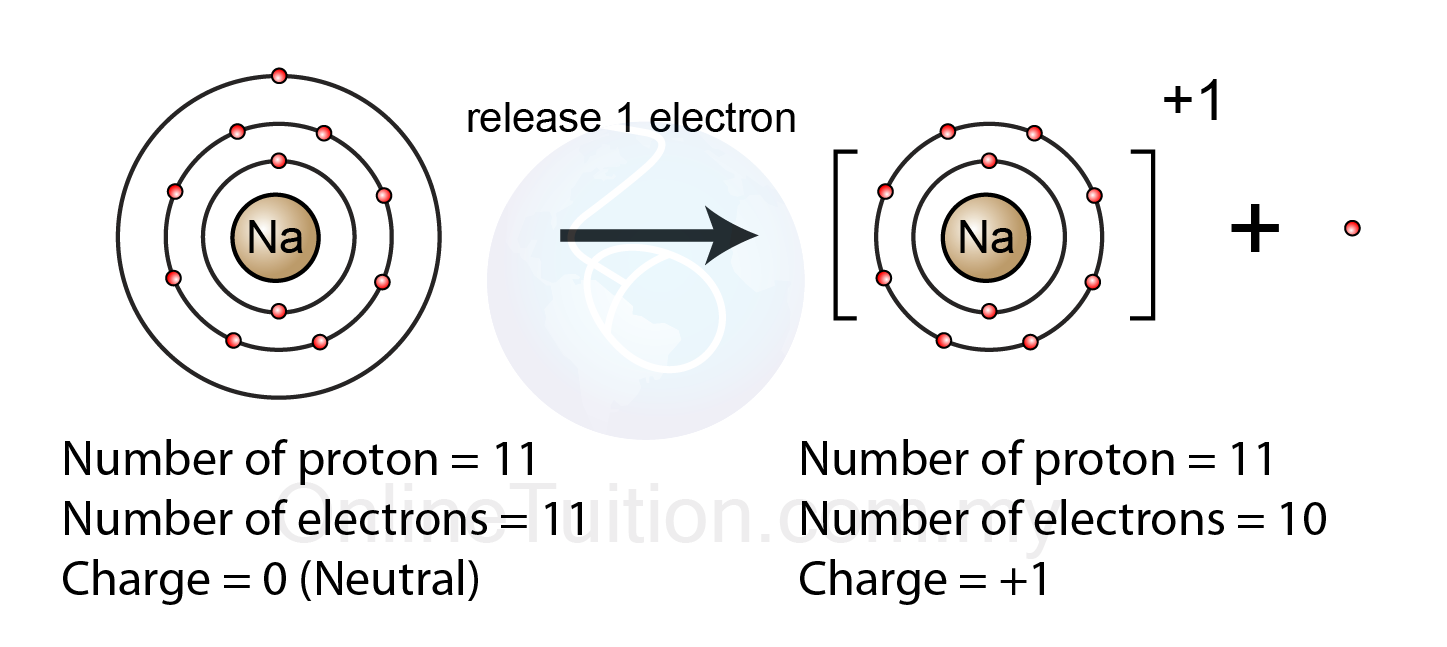
Again, principal shells with larger values of n lie at successively greater distances from the nucleus. Thus, the electron shells of an atom are populated from the inside out, with electrons filling up the low-energy shells closer to the nucleus before they move into the higher-energy shells further out. Explains that sodium and oxide ions have the same numbers of electrons, as Na has. When these two elements react, sodium gives the one electron in. The reason is the same as for atomic radii: shielding by filled inner shells produces little change in the effective nuclear charge felt by the outermost electrons. sodium and oxygen in terms of electron arrangement and atomic structure. Chlorine, on the other hand, has an atomic number of 17 and has 7 electrons in its outer shell. Ionic radii follow the same vertical trend as atomic radii that is, for ions with the same charge, the ionic radius increases going down a column. To save room, the configurations are in noble gas shorthand. 
This list of electron configurations of elements contains all the elements in increasing order of atomic number. Shannon, “Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides,” Acta Crystallographica 32, no. The electron configuration shows the distribution of electrons into subshells. Name: Sodium Atomic Number: 11 Element Symbol: Na Group: 1 Period: 3 Block: s Element Family: alkali metal Atomic Mass: 22.989 769 28(2) Electron Configuration: Ne3s 1 (shorthand) or 1s 2 2s 2 2p 6 3s 1 (full) Discovery: Sir Humphry Davy in 1807 Davy isolated sodium from sodium hydroxide using electrolysis. \): Ionic Radii (in Picometers) of the Most Common Ionic States of the s-, p-, and d-Block Elements.Gray circles indicate the sizes of the ions shown colored circles indicate the sizes of the neutral atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
